Reduced or no-energy output during HV therapy in ICDs/CRT-Ds manufactured with a specific (glassed) feedthrough, including currently available ICDs and CRT-Ds.
ANZCDACC Hazard Alert and Product Defect Correction May 2023
Device:
Cobalt™ XT/Cobalt™/Crome™ ICDs and CRT-Ds
A subset of: Claria MRI™/Amplia MRI™/Compia MRI™/Viva™/Brava™ CRT-Ds
A subset of: Visia AF™/Visia AF MRI™/Evera™/Evera MRI™/Primo MRI™/Mirro MRI™ ICDs
TGA Reference:
RC-2023-RN-00433-1
Australian Register of Therapeutic Goods (ARTG):
281611, 230019, 281610, 230021, 339481, 339487, 339482, 339489, 339483, 339490, Cancelled: 203212, Cancelled: 219221, 281607, 280186, 281608, 280183, 281606, 280184, 280182, 341548, 341556, 341553, 341549, 341551, 341557, 341554, 341550, 341552, 341555, 341547, 282960, 280354, 282961, 280355, 281609, 230020, 339484, 339491, 339485, 339488, 339486, 339492
Wand Notification Reference:
|31099
Web Assisted Notification of Devices (WANDS):
160907-WAND-6MO71M, 140408-WAND-6HEYLA, 160907-WAND-6MO6Y6, 140408-WAND-6HEYM7, 200109-WAND-6TXFRQ, 200109-WAND-6TXFST, 200109-WAND-6TXFSH, 200109-WAND-6TXFT3, 200109-WAND-6TXFVH, 200109-WAND-6TXG6G, Cancelled: 120823-WAND-6DVT1J, Cancelled: 130925-WAND-6G963U, 160901-WAND-6MMWH3, 160225-WAND-6LIENL, 160901-WAND-6MMWKA, 160225-WAND-6LIEMU, 160901-WAND-6MMWBT, 160225-WAND-6LIEL8, 160225-WAND-6LIEJE, 200109-WAND-6TXFO3, 200109-WAND-6TXFOZ, 200109-WAND-6TXFN0, 200109-WAND-6TXFJ5, 200109-WAND-6TXFOK, 200109-WAND-6TXFP9, 200109-WAND-6TXFNA, 200109-WAND-6TXFKX, 200109-WAND-6TXFQB, 200109-WAND-6TXFQ1, 200109-WAND-6TXFPN, 160908-WAND-6MOCJD, 151022-WAND-6KRD11, 160908-WAND-6MOCLI, 151022-WAND-6KRD1N, 160907-WAND-6MO6X8, 140408-WAND-6HEYN9, 200109-WAND-6TXFTE, 200109-WAND-6TXFTO, 200109-WAND-6TXFTX, 200109-WAND-6TXFU9, 200109-WAND-6TXG78, 200109-WAND-6TXG7J
Advisory grade TGA: Class I
(Class I recall action occurs when the product deficiency is potentially life-threatening or could cause a serious risk of health).
Description:
There is a rare potential for reduced or no-energy output during high voltage (HV) therapy (typically 0-12J) in implantable cardioverter defibrillators (ICDs) and cardiac resynchronisation therapy defibrillators (CRT-Ds) manufactured with a specific (glassed) feedthrough, including currently available ICDs and CRT-Ds.
There is an increased potential for a reduced or no-energy HV therapy in the AX>B configuration when all the following conditions are met:
- The device has a glassed feedthrough (manufactured after July 2017).
- There is significant separation of the layers of insulation materials in the feedthrough components of the device header.
- An unintended current pathway forms within the void created by the insulation separation, capable of conducting high levels of current during HV therapy.
When an unintended current pathway is detected during HV therapy, the Short Circuit Protection (SCP) feature may trigger. SCP is a safety feature that can only occur during high voltage (HV) therapy. SCP is designed to truncate energy delivery to protect the device when an unintended current pathway is detected during a shock. This behaviour can be intermittent; both full-energy and reduced-energy HV therapies within the same episode have been observed. SCP events may also be lead related; for both lead-related and device-related unintended current pathways, the defibrillation waveform is truncated early in the energy delivery sequence, resulting in reduced or no energy being delivered (~0-12J).
There have been no deaths due to this issue in the glassed feedthrough device population, although there have been instances where external defibrillation was required to convert an arrhythmia, due to failure of the device to deliver adequate therapy.
In some cases, a persistent 50% drop in all pacing lead impedances may be displayed; however, lead function, is not affected.
Should this issue occur, pacing, sensing, episode detection, anti-tachycardia pacing (ATP) therapies, battery longevity, and telemetry remain functional.
Medtronic is updating Instructions for Use, HV therapy nominals and the programmer interfaces for these device models to be consistent with information in this report. Medtronic will release additional information once the necessary regulatory approvals have been received, as applicable in the local region.
The root cause for device related first-phase SCP events, the sequence of events and the physics of failure are still being investigated by Medtronic.
Number of devices affected in Australia and New Zealand:
Not supplied
Presentation:
As a result of shocks below the defibrillation threshold (DFT) and subsequent delay/absence of receiving successful shocks to terminate ventricular arrhythmia, patients affected by this issue may present with symptoms of hemodynamic compromise, multiple shocks and potentially sudden cardiac death.
The potential harm to the patient could also include complications associated with device replacement and/or unnecessary lead replacement if the reduced- or no-energy HV therapy is erroneously attributed to a lead failure.
Upon interrogation, the device will report in the “Episode Text” the delivered energy as less than the programmed energy, as shown in the example image below.
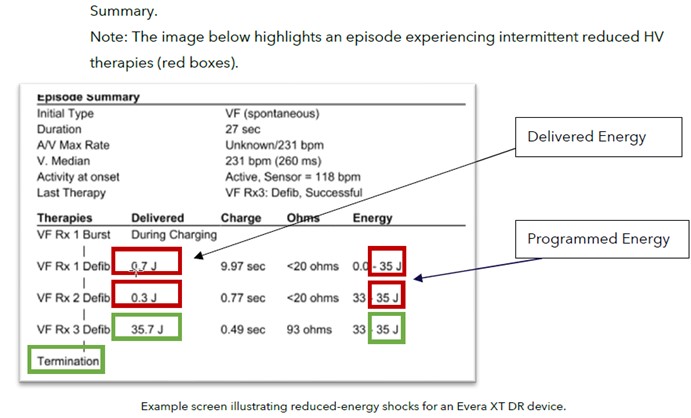
Rate of occurrence:
Through 10 April 2023, Medtronic has identified 27 devices from approximately 816,000 devices (representing 0.003%) distributed worldwide that have experienced a reduced or no-energy HV therapy due to the issue described above. Of these, 26 were in devices with an AX>B delivered pathway.
Based on an analysis of patients with a glassed feedthrough device and with a history of HV therapy, the observed rate for this issue is 0.03%, highlighting the observed failure rate would be much higher if all devices were required to deliver therapy.
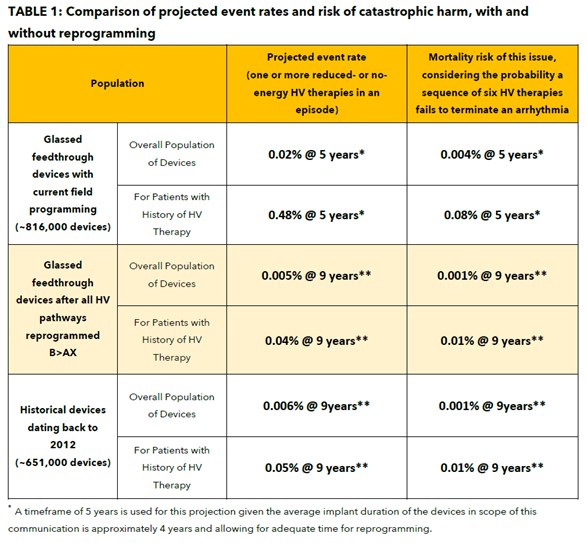
With current field programming, devices with a glassed feedthrough may experience increased risk for reduced or no-energy output during HV therapy, projected at 5 years to be 0.02% overall and 0.48% for patients with a history of HV therapy.
Medtronic conducted a broader analysis to determine the incidence of device related reduced or no-energy HV therapy events outside of the above population, with implants dating back to 2012. Using these historical observed events, projected rates for this population of devices is 0.002% at five (5) years and 0.006% at nine (9) years. This analysis identified two deaths due to this issue in the brazed feedthrough device population suggesting a device-related reduced or no-energy HV therapy occurred.
When devices in the glassed feedthrough population are programmed exclusively in the B>AX configuration, the overall potential for a reduced or no-energy HV therapy is 0.002% at five (5) years and 0.005% at nine (9) years, comparable to historical device performance.
Recommendation:
Individual devices susceptible to this issue can be identified via search/look-up on the Medtronic Product Performance Report Website (http://productperformance.medtronic.com).
- Prophylactic device replacement is NOT recommended.
- The risk of mortality for patients after reprogramming is 0.001% at 9 years and is less than the risk of patient mortality due to complications associated with device replacement (0.032% – 0.043% 1,2,3).
- Program all HV therapy pathways B>AX in all therapy zones to minimise the risk for this issue.
- Note: Using “Get Medtronic Nominals” will require manual reprogramming of Rx5 and Rx6 to B>AX for all ventricular therapies.
- Prioritise reprogramming patients who are at the highest risk:
- Namely those who have a history of HV therapy and/or secondary prevention indication AND Rx1 programmed AX>B.
- Rx1 provides the greatest statistical likelihood to resolve an arrhythmia, and therefore it is important to minimise risk of a reduced or no-energy HV therapy in the first sequence.
- Ask your local Medtronic Representative to arrange a search for these high-risk patients on Carelink.
- For remaining patients with AX>B programming in any HV therapy sequence, schedule (with appropriate discretion) the next follow-up for in-clinic reprogramming to minimise potential for reduced or no-energy HV therapies to occur.
- Per standard practice, check tachyarrhythmia episodes to determine effectiveness of therapies that have been delivered.
- Instruct patients to contact the clinic if they receive HV therapy or hear an audible tone coming from their device.
- Verify delivered energy is consistent with programmed energy in the Episode
- Contact your local representative if one of the following is observed as these may be an indication of either a device or lead-related issue:
- Reduced- or no-energy HV therapy is displayed in Episode Text (regardless of programmed pathway)
- A persistent drop of approximately 50% in RA, RV and LV pacing lead impedance measurements as this may be an indication of increased potential for a future reduced- or no-energy therapy.
- Namely those who have a history of HV therapy and/or secondary prevention indication AND Rx1 programmed AX>B.
- Tarakji KG, et al. Antibacterial Envelope to Prevent Cardiac Implantable Device Infection. The New England Journal of Medicine. 2019; 380(20):1895-1905.
- Medtronic Data on File. MDT2260884-CRHF CIED Infection Report; Agile: MDT2260884, Version 2.0, 11/02/2015.
- Birnie D, et al. Complications associated with defibrillation threshold testing: The Canadian experience. Heart Rhythm. 2008; 5(3):387-90.
The ANZCDACC encourage you to report any adverse event or near (potential) adverse event associated with the use of a medical device including any abnormal CIED or lead function. We encourage reporting to ANZCDACC directly via the Committee chair Dr Paul Gould [email protected] and to the following regulators.
In Australia, report to the TGA;
| Online | https://www.tga.gov.au/reporting-problems |
In New Zealand, report to Medsafe;
| Post | Compliance Management Branch, Medsafe, PO Box 5013, Wellington 6145. |
| [email protected] | |
| Fax | 04 819 6806 |
